
The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. Likewise, bromine will have a larger atomic radius when compared with chlorine. Which element in Period 2 has the largest atomic mass It is clear that the element with most mass in Period 2 is Ne, which has an atomic number of 10 and a molecular weight of 21. The cookie is used to store the user consent for the cookies in the category "Performance". The element in the second period that has the largest atomic radius is the Lithium (Li). Thus, helium is the smallest element, and francium is the largest. As can be seen in the figures below, the atomic radius increases from top to bottom in a group, and decreases from left to right across a period. This cookie is set by GDPR Cookie Consent plugin. Atomic radii vary in a predictable way across the periodic table. The cookie is used to store the user consent for the cookies in the category "Other. Thus, the greatest amount of energy is required to remove an electron from chlorine atom. Measuring Atomic Radius The atomic radius of a particular element is an important characteristic as it helps us to understand many properties of atoms and how they react. 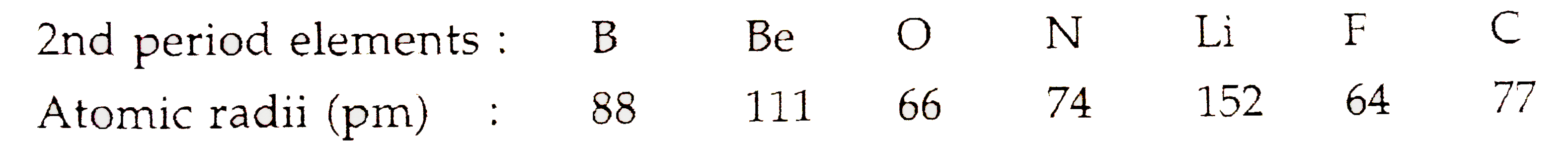
This cookie is set by GDPR Cookie Consent plugin. Helium is in the top period and the farthest right group, which follows the patterns of atomic radius on the periodic table. The cookies is used to store the user consent for the cookies in the category "Necessary". This cookie is set by GDPR Cookie Consent plugin. The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Namely, this is due to the fact that calciums. Meanwhile, all the elements including N and Li in the second period has the same number of electron energy level (n=2), so shielding effect is the same and does not affect the judgement.Necessary cookies are absolutely essential for the website to function properly. Because calcium is located closest to the bottom right on the periodic table, it has the highest atomic radius. Reason is, the increasing nuclear charge (N has 7 proton while Li has 3 proton) pulled back the electrons with a greater power, making the atomic size of nitrogen smaller. 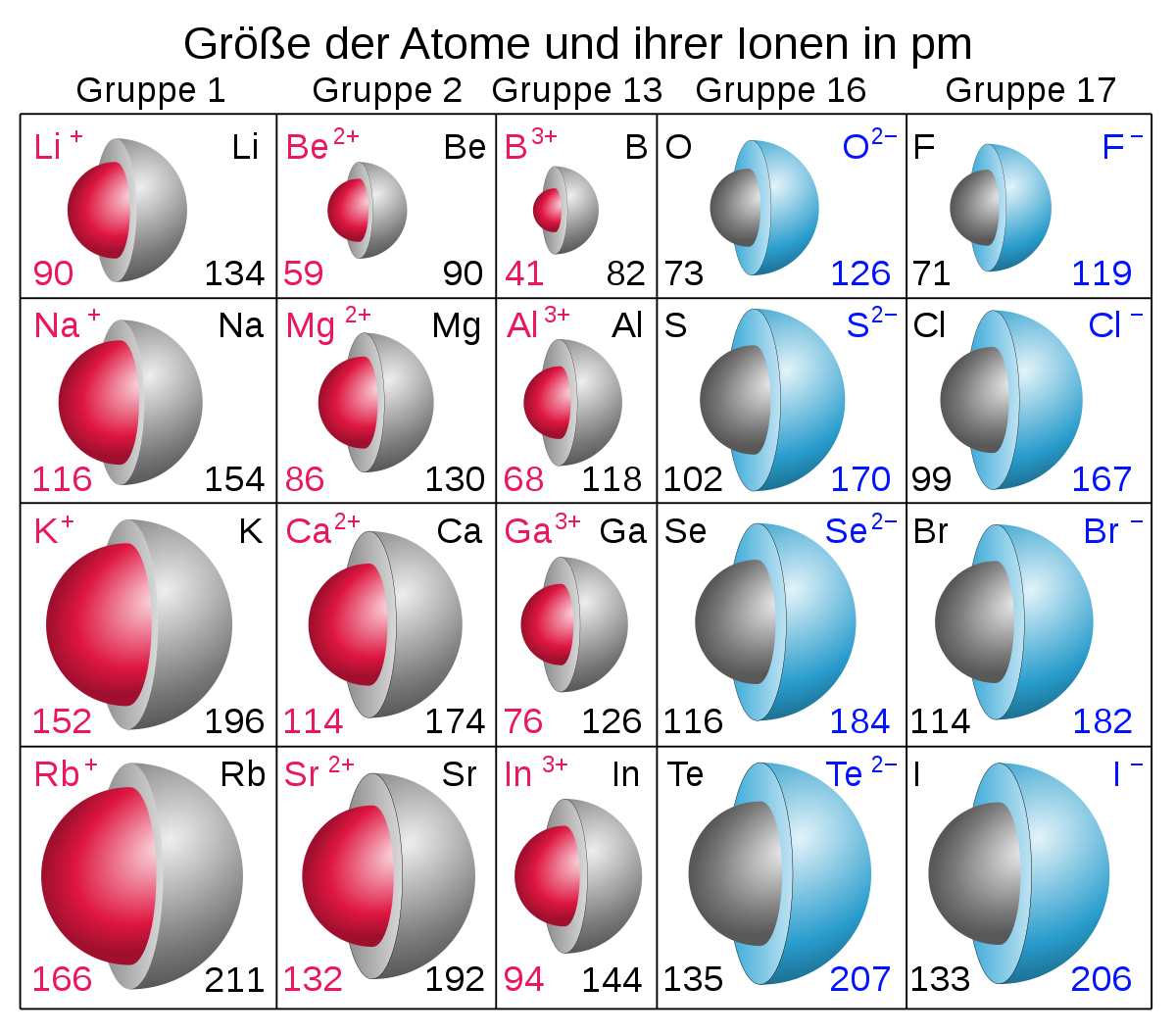
For example, in the second period, the atomic radius of Nitrogen (N) is smaller than that of Lithium (Li). K has the largest atomic radii because atomic radii decrease from left to right along a period. And since the size of atom is mostly consist of electrons, more energy level is filled, the larger the atom will be.)Ģ) Atom size (Atomic radius) decreases from left to right along each period of the periodic table. cobalt 5.which among the elements has the lowest metallic property a. Ra, Ba, Sr, Ca 4.which among the elements has the smallest atomic radius a. Reason is, the increasing energy level (increases from n=1 to n=7) causes the shielding effect increase dramatically. 3.which group of elements bellow follows an increasing trend a. For example, in the first group, the Atomic radius trend from smallest to greatest is: H (smallest) - Li - Na - K - Rb - Cs - Fr (greatest). 1) Atom size (Atomic radius) increases from up to down along each column(group) of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
